New Syringe Label with First-Opening Indication from Schreiner MediPharm Increases Patient Safety
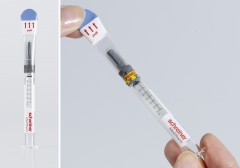
In many daily healthcare situations, every second counts. The fewer the moves staff have to make the better for the patient. Consequently, prefilled syringes are frequently used for injections. Schreiner MediPharm has developed a user-friendly security label for sealing prefilled syringes enabling healthcare professionals to quickly tell that the drug they are about to administer is an original that has not been opened before or tampered with: The Syringe-Closure-Wrap delivers clear first-opening indication of the label with one flick of the wrist, protects the integrity of prefilled syringes and thus enhances patient safety.
The innovative Syringe-Closure-Wrap marking solution features a label that wraps around the syringe barrel and the complete cap. When the user pulls off the upper part of the label, the integrated first-opening indication is triggered. Due to special security die cuts, the label is irreversibly destroyed and cannot be closed again unnoticed. After the label has been opened for the first time, an additional warning appears as a visible security element. For increased product and counterfeiting protection, overt, covert or digital authentication features can be integrated into the label.
The Syringe-Closure-Wrap is adaptable to all commonly used syringe sizes. In contrast to shrink-wrap solutions, no direct heat is applied in processing the syringe label, which makes it suitable also for heat-sensitive substances. The enlarged label area extending across the cap can be used for additional information so that the compact label design offers ample space for clearly marking the product while protecting the integrity of the syringe.
Schreiner MediPharm’s novel Syringe-Closure-Wrap for sealing prefilled syringes enables healthcare professionals to effectively detect at a glance if a medication has been previously opened or tampered with. As a result, patients can be reliably protected against the administration of potentially counterfeit or contaminated products.